To provide you with the most complete findings from whole genome sequencing tests for neurological conditions, a new gene panel is being created that will be used across multiple clinical indications: "Adult-onset neurological disorders"
Urgent advice: Upcoming changes
The below changes take effect following publication of the updated NGTD by NHS England and once the new WGS panel is implemented in Panel App, expected in May 2026. We aim to provide efficient implementation when this happens. This post will be updated when the changes are in effect.
The new panel will be used across the tests listed below and brings together several existing overlapping gene panels. This means reports will contain results based on analysis of a larger number of genes and will reduce the need to request reanalysis.
These changes will be introduced as part of an upcoming updates to the national genomic test directory (NGTD) and PanelApp. To receive updates on our services, including NGTD changes please sign up to our mailing list.
Overall, these changes will replace one existing clinical indication (R58) with four new ones and will introduce a new single gene panel that will be used across the four new indications and three other existing clinical indications to deliver one conclusive genetic result without the need for further testing.
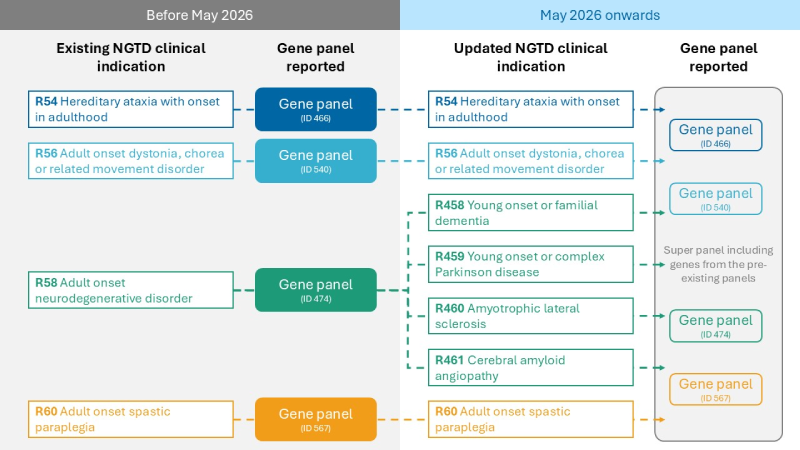
Dividing adult-onset neurodegeneration (R58) into new clinical indications
In the updated NGTD, the test for adult-onset neurodegeneration (R58) will be replaced with four new, more specific clinical indications. This change is expected to improve clarity in communicating with the lab and ensure results are interpreted in a way that is most relevant to your clinical practice.
Please use the new clinical indications below in place of R58:
- R458 Young onset or familial dementia
- R459 Young onset or complex Parkinson disease
- R460 Amyotrophic lateral sclerosis / Motor Neurone Disease
- R461 Cerebral amyloid angiopathy
In the short term, where sufficient clinical information is provided, we will automatically reassign orders for R58 to the most appropriate of the new clinical indications. Where this is not possible, we will contact you for guidance.
Neurological clinical indications included in the new panel
The new single neurological gene panel [Adult-onset Neurological Disorders] will be used for analysis and reporting across seven WGS tests. The clinical indications that will use the new panel are:
- R54 Hereditary ataxia with onset in adulthood
- R56 Adult-onset dystonia, chorea or related movement disorder
- R60 Adult-onset hereditary spastic paraplegia
- R458, R459, R460, R461 The four new clinical indications that have replaced R58
All genes previously analysed through any of these tests will be included in the new panel and will be covered in the resulting report.
Ordering any one of these tests will provide comprehensive results relevant to all of these clinical indications. Do not request re-analysis under another one of these clinical indications.
Please consult the full eligibility criteria in the national genomic test directory when ordering tests.
These changes will be part of a wider update to the national genomic test directory.
If you have any questions, please contact us.

