Whole Genome Sequencing (WGS) is available through the NHS Genomic Medicine Service for all patients aged 25 and under with confirmed or suspected cancer, or sarcoma of any age.
Urgent advice: On this page
- Key benefits of Whole Genome Sequencing for Cancer
- Eligibility: Who should be considered for WGS?
- Tools and resources
- Multidisciplinary Teams and Genomics Community of Practice
- Consent and Pre-test discussion
- Ordering a Solid Cancer WGS test: Referral process, guidance for completing a WGS test order form, RNAlater (samples not requiring freezing)
- Test Turnaround Times
- Interpreting test results and integration into care
- Cancer WGS FAQs
- Patient information and support
WGS analyses tumour and germline (blood/saliva) samples to guide diagnosis and treatment, often identifying rare, actionable mutations for TYA (Teenage and Young Adult) patients.
Key aspects of WGS for TYA Cancer:
- Procedure: Requires both a tumour sample and a germline sample (blood, saliva, or skin) to compare against.
- Purpose: Helps determine precise treatment options, identifies cancer-causing variants, and checks for inherited cancer risks.
- Consent: Specialized, dedicated consent is required for WGS, often involving a Record of Discussion (RoD) form.
- Turnaround: WGS can confirm rare, or unexpected diagnoses, improving treatment precision for younger patients.
Eligibility: Who should be considered for WGS?
The eligibility criteria in the NHS in England is as follows:
- Any patient <25 years of age with a suspected or confirmed malignant tumour (solid cancer, Cancer of Unknown Primary (CUP), ovarian cancer, triple-negative breast cancer, or blood cancer), along with specific, rare adult cancers (e.g., sarcomas).
- Rare tumour types or those typical of paediatric presentations occurring in young adults.
- Cases where standard molecular testing is insufficient or treatment options are exhausted.
- Sarcomas and CNS cancers often flagged for sequencing regardless of age because of genomic complexity.
Please see eligibility criteria as outlined in the National Genomic Test Directory (opens in a new tab).
Multidisciplinary Teams and peer support
The decision to request WGS should ideally be reviewed in a multidisciplinary team (MDT) context alongside radiology, pathology and oncology input.
East Midlands TYA MDT Meetings
- Held weekly on a Friday between 12 –1pm, and you can join the MDT remotely via video link.
- Referrals for discussion at the TYA MDT are to be received no later than 4pm on a Wednesday to be included on that week’s Friday agenda. Any referrals received after this will be discussed at the MDT the following week.
- All referral forms are to be submitted and sent via secure NHS.net email to NUHNT.EMTYAMDT@nhs.net
- Ideally patients should be referred to the TYA MDT within 14 days of diagnosis and where this is not possible, they should be referred into the service within 28 days.
For advice and support regarding the correct pathway, please contact the East Midlands TYA team on nuhnt.teenageandyoungadultteam@nhs.net
East of England TYA MDT Meetings
- All TYA patients in the region with a confirmed cancer diagnosis should be referred to the TYA Cancer MDT meeting at the Principal Treatment centre for TYA's with cancer, Addenbrooke’s Hospital.
- Referrals for discussion at the TYA MDT, which is held weekly on a Monday between 11am and 12.15pm, are to be received no later than 2pm on a Friday in order to be included on that week’s agenda. Any referrals received after this will be discussed at the MDT the following week.
- All referral forms are to be submitted and sent via secure NHS.net email to add-tr.cancermdt@nhs.net
For advice and support regarding the correct pathway, please contact the East of England TYA team on cuh.tyateam@nhs.net (Telephone: 01223 274243 / 349270).
Further support for clinicians in the East of England is available from the East of England Cancer Alliance Genomic Practitioner Service.
Informed Consent & Pre-Test Discussion
Clinicians must have a detailed conversation with the patient (and where appropriate families) about:
- What WGS can and cannot find (somatic drivers vs incidental or secondary findings).
- The possibility of germline pathogenic variants that could affect cancer risk and family members.
- Options around research use and sharing of genomic data (e.g., national research libraries).
- Whether the patient wants to be informed about secondary findings (variants unrelated to their current cancer but medically significant).
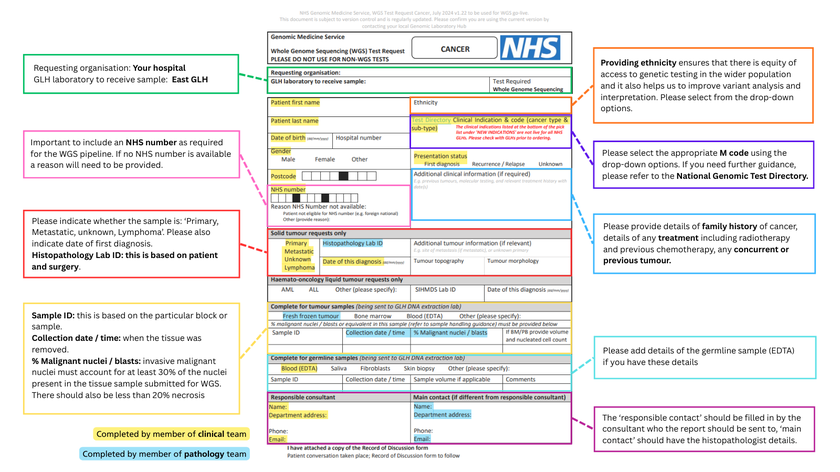
Ordering a Solid Cancer WGS test
To order a WGS test, please refer to our Cancer test web page (also relevant for heamatological malignancies).
As well as completing the order form and record of discussion form (which is available in a number of languages), other forms may be necessary depending on the age of the patient or if they lack capacity. These are listed on the webpage links provided.
Please ensure the correct order forms are used.
Referral process
Non-urgent advice: Did you know? You can now send solid cancer samples to East Genomics using RNAlater.
RNAlater removes the need for immediate freezing or dry ice shipment in many cases and makes out-of-hours sampling safer and more practical.
The introduction of RNAlater provides an alternative to the collection and storage of fresh-frozen samples in situations where this is impractical or infeasible.
Find out more about the RNAlater sampling pathway, including how to order kits, on our RNAlater sampling page.
Guidance for completing a WGS (Solid Cancer) Test Order Form
The WGS requesting form is available here (opens in a new tab) (see Appendix for details on navigating the auto populated form).
Test Turnaround Times
See our NHSE Test Directory turnaround times page for further details.
For further information about when to expect the results of a requested test, please contact our labs.
Interpretating results and integration into care
Combining WGS with clinical data gives the best context for interpretation.
Somatic results can:
- Identify actionable genetic drivers or targets for targeted therapies or clinical trials.
- Refine diagnosis and classification of cancers with atypical features.
- Inform prognostic stratification in some cancers.
Germline findings:
- May indicate hereditary cancer syndromes - referrals to genetic counselling are often needed.
- Need careful communication regarding family risk, insurer implications (legislation varies), and long-term follow-up.
Variant interpretation:
- Be prepared for variants of unknown significance (VUS); these may require specialist review and periodic re-interpretation as evidence evolves.

